Status
Completed
Period
15 January – 15 March 2023
Applicant
Beatriz Leiva Jiménez
Home Institution
BCMaterials – Basque Center for Materials, Applications and Nanostructures
Host Contact
Dr. Peter Van Oostrum
Host Institution
Department of Bionanosciences (DBNS). University of Natural Resources and Life Sciences (BOKU)
Aim of the mission
During of STSM, concept of protein translocation by shear forces though functionalized brushes into nanopores has been explored. This phenomena can provide a solution to protein agglomeration and unfolded states, where the function of the protein is limited. The main goal was studying the refolding process on knotted proteins and compare the behaviour to unknotted proteins.
Summary of the Results
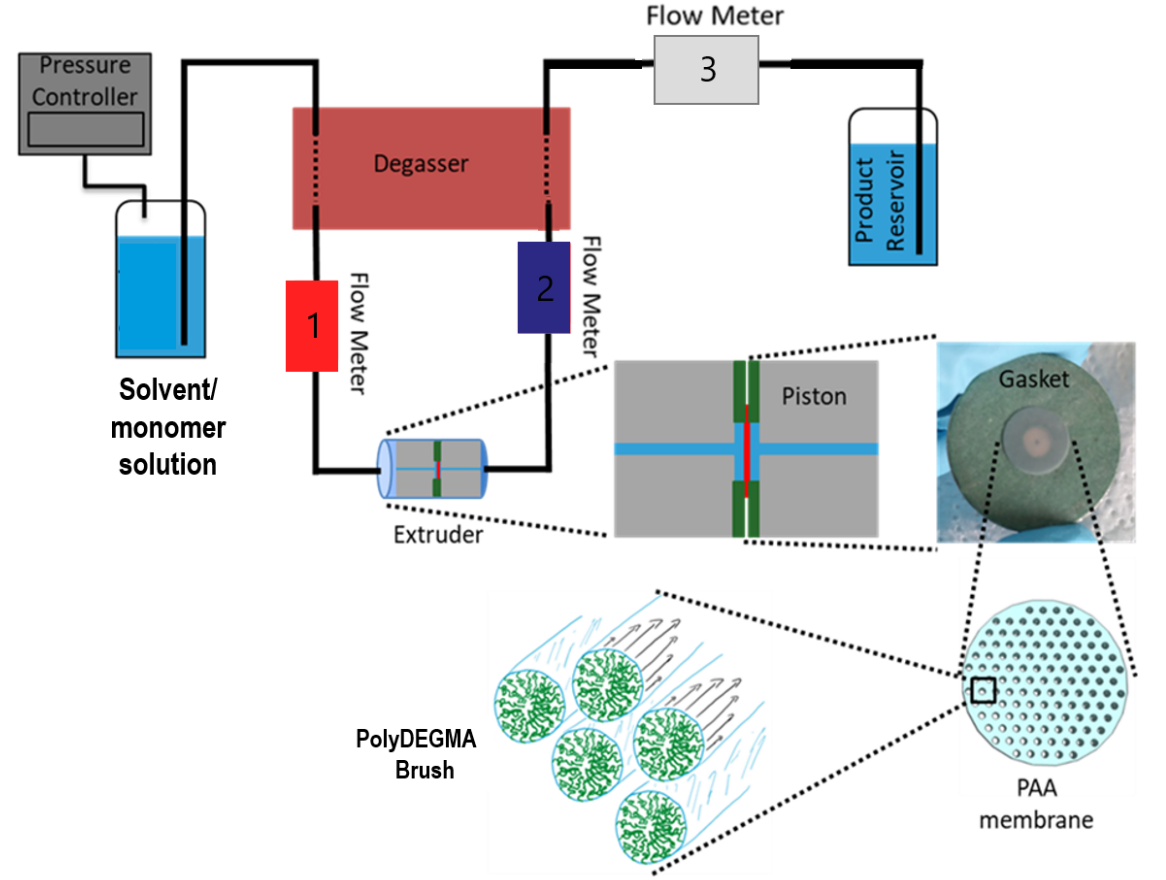
The polymerization of DEGMA monomers into the nanopores for the synthesis of polyDEGMA brushes were successfully performed by opening-ring chemistry (ATRP). After that, characterization assays for testing the brush behaviour were established (flow resistant analysis and SEM) keeping in mind some parameters as temperature and the type of solvent. These can control the hydration status of the brush (and consequenty, its functionality) during protein extrusion. For flow resistance analysis, a temperature sweep was carried out in a range where proteins are not denatured in two different solvents, miliQ water and PBS. They were extruded through the chip from 4 to 45ºC, and then back to 4ºC. The results showed a similar shape in both solvents, confirming the reproducibility of the experiment. Futhermore, this also helped understanding the hydration status of the brush by the transition temperature (TT), which is the degree where the brushes start to dehydrate. Protein assays were performed 5ºC below that the TT for ensuring the good brush hydration status. For analysing protein folding by shear forces and disaggregation process, a model protein as BSA was extruded for analysing the brush behaviour before and after the extrusion. The decrease of flow resistance while BSA extrusion revealed the good functionality of the brush. After that, BSA was replaced forlysozyme in is natured and denatured form for studying the brush ability for refolding proteins. In first place, the activity of natured lysozyme before extrusion was tested for comparing the after extrusion one. In second place, the activity of the denatured one was also analysed before and after extrusion. In some future experiments, denatured GFP will be extruded while a UVvis adsorption measurement is performed in order to analyse its translocation into a functional protein, so fluorescence signal will be revealed.
Dissemination
The planned aims of this project were successfully achieved during this STSM. However, more investigation is required. New outcomes are expected and also, a publication based on the obtained results. A collaboration between BOKU and BCMaterials is now established, which will allow understanding better the refolding principle through translocation. This was predicted by computational models in Spain and applied in Austria. Thanks to STSM, a new microfluidics system will be established in Spain which will help achieving the planned outcomes. For now, the optimization of the microfluidics system will be part of the short- term planned goals, considering the application of different functionalization parameters such as different polymerization temperature and monomer percentage, which will allow a slow monomer functionalization that can be controlled, reproducible and, eventually, scalable at industries.