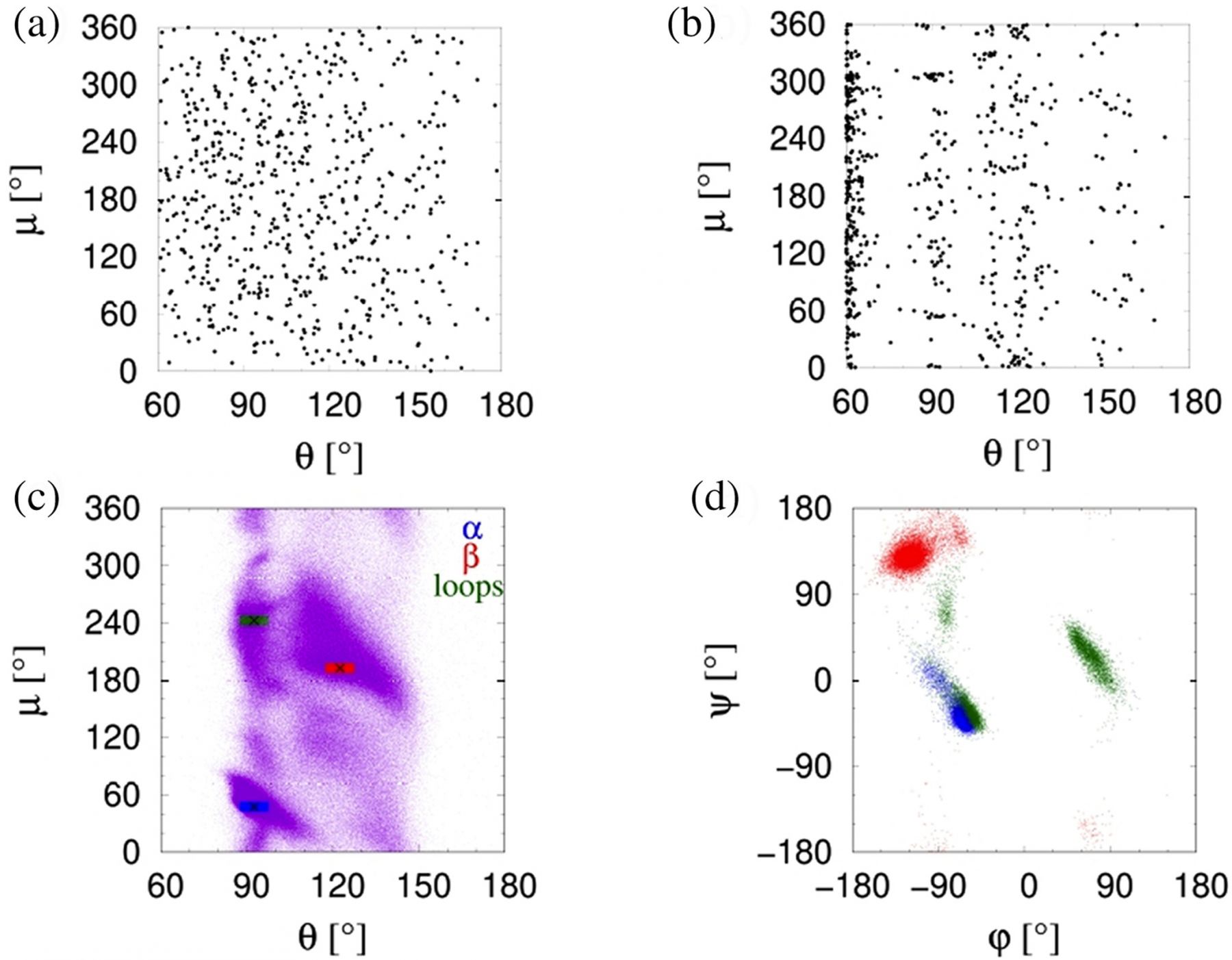
We seek to understand the interplay between amino acid sequence and local structure in proteins. Are some amino acids unique in their ability to fit harmoniously into certain local structures? What is the role of sequence in sculpting the putative native state folds from myriad possible conformations? In order to address these questions, we represent the local structure of each C-alpha atom of a protein by just two angles, theta and mu, and we analyze a set of more than 4000 protein structures from the PDB. We use a hierarchical clustering scheme to divide the 20 amino acids into six distinct groups based on their similarity to each other in fitting local structural space. We present the results of a detailed analysis of patterns of amino acid specificity in adopting local structural conformations and show that the sequence-structure correlation is not very strong compared to a random assignment of sequence to structure. Yet, our analysis may be useful to determine an effective scoring rubric for quantifying the match of an amino acid to its putative local structure.
Download a copy of the manuscript (preprint version)